Table of Contents
Affiliate link notice: As an affiliate of BetterHelp and other third-party vendors, we may receive compensation if you make a purchase using the links provided on this page. For more information, visit our disclosure page.
Last Updated on March 2, 2026 by Randy Withers
Some people are treated for anxiety for years before anyone asks what they’re eating.
They describe chronic tension, irritability, brain fog, low energy, disrupted sleep — and somewhere in the background, persistent bloating, unpredictable digestion, or abdominal discomfort that gets dismissed as “just IBS.” The mind is treated. The gut is tolerated. And the two are rarely considered together.
The reality is that the gut and the brain are in constant conversation. Through a complex network of neural pathways, immune signaling, hormones, and microbial metabolites — collectively known as the gut–brain axis — these systems regulate one another in ways that are both measurable and clinically significant. The gut contains roughly 500 million neurons. It produces the vast majority of the body’s serotonin. While peripheral serotonin does not directly cross into the brain, the biochemical activity of the digestive system influences inflammation, vagal tone, and stress signaling in ways that shape mood, cognition, and emotional regulation.
When the digestive system is chronically irritated — particularly in conditions such as FODMAP Sensitivity — the effects do not remain confined to the abdomen. Repeated fermentation, immune activation, altered motility, and visceral hypersensitivity create a state of physiological stress that the nervous system registers and responds to. Over time, that feedback loop can amplify anxiety, destabilize mood, and impair cognitive clarity.
This article examines how that process unfolds. We will look at what the gut–brain axis actually does, why FODMAP Sensitivity can become a self-reinforcing gut–brain loop, how related intolerances may compound the problem, and what evidence-based interventions meaningfully restore stability. The goal is not dietary perfection. It is physiological clarity — because before assuming your brain is malfunctioning, it may be worth asking whether your gut has been under chronic strain.

What the Gut–Brain Axis Actually Does
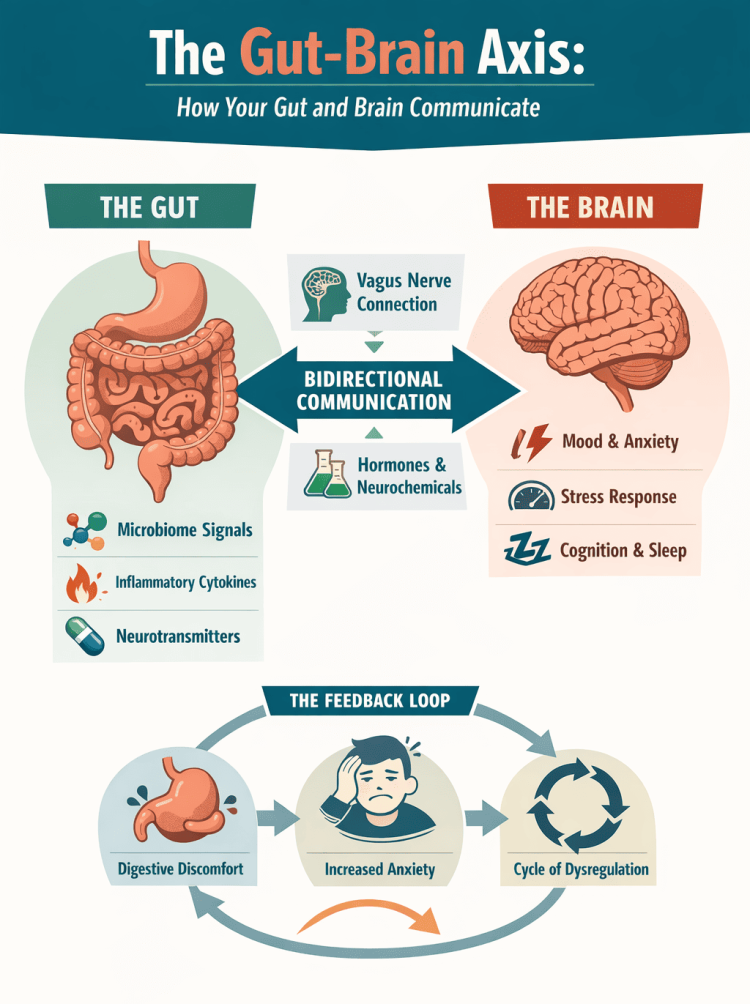
The gut–brain axis is not a metaphor. It is a bidirectional communication system linking the central nervous system and the enteric nervous system. The vagus nerve, running from the brainstem into the abdomen, serves as one of its primary conduits, transmitting signals in both directions. The gut sends information upward about nutrient composition, microbial metabolites, immune activation, and mechanical distension. The brain sends signals downward regulating motility, secretion, blood flow, and inflammatory responses.
When functioning properly, this system maintains equilibrium. Digestion proceeds efficiently. Immune responses remain proportionate. Stress responses resolve rather than linger. The microbiome produces short-chain fatty acids that reinforce gut lining integrity and modulate inflammation.
When the system becomes dysregulated, the consequences extend beyond discomfort. Chronic gut inflammation can elevate pro-inflammatory cytokines that influence central nervous system activity. Alterations in the microbiome may affect neurotransmitter precursors and vagal signaling. Visceral hypersensitivity — common in IBS — lowers the threshold at which normal digestive processes are perceived as painful. What might be minor gas expansion in one person becomes sharp cramping in another.
Importantly, this heightened sensitivity is physiological, not psychological. The nerves lining the gastrointestinal tract send amplified signals to the brain. The brain, in turn, responds with heightened vigilance and stress activation. That reciprocal signaling can create a loop: discomfort increases anxiety; anxiety increases gut reactivity; gut reactivity reinforces discomfort.
Food intolerances sit squarely within this feedback system. When specific carbohydrates or compounds are poorly absorbed, fermented, or immunologically provocative, the gut does not merely experience transient irritation. It enters a repeated cycle of fermentation, inflammation, and neural signaling that the brain cannot ignore.
Nowhere is this dynamic more clearly illustrated than in FODMAP Sensitivity.
FODMAP Sensitivity and the Gut–Brain Feedback Loop
FODMAP Sensitivity is one of the most common — and most frequently misunderstood — drivers of gut–brain dysregulation.
FODMAPs are short-chain fermentable carbohydrates found in everyday foods such as garlic, onion, wheat, apples, legumes, and certain dairy products. In individuals with irritable bowel syndrome (IBS) or heightened gut sensitivity, these compounds are incompletely absorbed in the small intestine. They pass into the colon, where gut bacteria ferment them, producing gas and drawing water into the bowel. The result can be bloating, distension, cramping, and altered motility.
The digestive mechanics are well documented. The neurological implications are less commonly discussed.
People with IBS — a condition closely associated with FODMAP Sensitivity — often exhibit visceral hypersensitivity. This means the nerves lining the gastrointestinal tract respond to normal stimuli with exaggerated signaling. What might be a mild sensation of fullness in one person becomes sharp discomfort in another. The signal traveling to the brain is amplified. Over time, the brain learns to anticipate that discomfort, increasing vigilance and stress reactivity around digestive sensations.
This is not a matter of imagination or emotional fragility. It is altered neural signaling within the gut–brain axis.
Once that loop is established, a feedback cycle can emerge. Fermentation increases gas and distension. Distension activates hypersensitive neural pathways. The brain registers threat. Stress hormones rise. Elevated stress further alters gut motility and sensitivity. The flare intensifies.
Anxiety and IBS are deeply intertwined for this reason. Psychological stress is a well-documented trigger for symptom flares, and symptom flares reliably increase anxiety. When dietary triggers remain unmanaged, this loop can become chronic — reinforcing both gastrointestinal and emotional instability.
Addressing FODMAP Sensitivity, therefore, is not purely about digestive comfort. It is about interrupting a neurophysiological cycle. Dietary modification remains the most evidence-based approach, particularly through a structured low-FODMAP elimination and reintroduction protocol. For some individuals, additional strategies — including targeted enzyme support or carefully selected gut health supplements — may expand dietary flexibility and reduce symptom burden.
But the broader point is this: unmanaged FODMAP Sensitivity can masquerade as generalized anxiety, mood volatility, or chronic stress intolerance. When the gut is persistently inflamed or overstimulated, the brain operates in a state of heightened reactivity. Treating the psychological symptoms without stabilizing the gut can leave a key driver unaddressed.
This does not mean every case of anxiety originates in the digestive tract. It does mean that for a meaningful subset of individuals, especially those with IBS, the gut–brain feedback loop deserves serious clinical attention.
Other Conditions That Can Mimic or Compound FODMAP Sensitivity
FODMAP Sensitivity is common, but it is not the only dietary mechanism capable of destabilizing the gut–brain axis. Several other intolerances operate through overlapping pathways and can either mimic or compound the same feedback loops.
Lactose intolerance is perhaps the most straightforward example. When lactase enzyme levels are insufficient, lactose reaches the colon undigested and undergoes bacterial fermentation — producing gas, distension, and discomfort in a pattern strikingly similar to high-FODMAP foods. Because lactose is itself a FODMAP (a disaccharide), symptoms often overlap. Chronic exposure to poorly tolerated dairy can sustain low-grade inflammation and neural activation that reinforces the same gut–brain signaling seen in broader FODMAP Sensitivity.
Fructose malabsorption functions similarly. When fructose absorption capacity is exceeded — whether due to high intake or individual variability — excess fructose ferments in the colon, generating gas and osmotic shifts. Given how pervasive fructose is in processed foods and fruit, some individuals live in a near-constant state of mild gastrointestinal irritation without recognizing the source. The resulting discomfort, bloating, and unpredictability can quietly reinforce anxiety around eating and digestive sensations.
Non-celiac gluten sensitivity remains more controversial. Unlike celiac disease, it does not produce consistent autoimmune markers or intestinal damage detectable on biopsy. Yet a subset of individuals report neurological and cognitive symptoms — brain fog, fatigue, mood disruption — that improve when gluten-containing foods are removed. Some research suggests that components in wheat beyond gluten itself, including fructans (which are FODMAPs), may account for part of this effect. In other words, what is attributed to “gluten sensitivity” may in some cases reflect underlying FODMAP Sensitivity or overlapping carbohydrate malabsorption.
Histamine intolerance operates through a different but related pathway. In this condition, the body’s ability to degrade dietary histamine is impaired. Aged cheeses, fermented foods, wine, and certain fish can trigger symptoms ranging from headaches and flushing to anxiety and sleep disturbance. Because histamine interacts directly with the nervous system, elevated levels can amplify both gastrointestinal and psychological symptoms, further complicating the gut–brain picture.
The common thread across these conditions is not simply digestive discomfort. It is repeated activation of inflammatory and neural signaling pathways that feed into the brain. When these exposures occur daily, the nervous system adapts accordingly — often toward heightened sensitivity.
For individuals already struggling with FODMAP Sensitivity, unrecognized overlapping intolerances can make progress feel inconsistent or incomplete. Precision matters. The more accurately dietary triggers are identified, the more effectively the gut–brain feedback loop can be interrupted.
What Actually Helps: How To Restore Gut–Brain Stability
The encouraging reality is that the gut–brain axis is responsive. The same bidirectional signaling that allows dysregulation to take hold also allows recalibration when meaningful changes are introduced. Improvement does not require perfection. It requires targeted intervention.
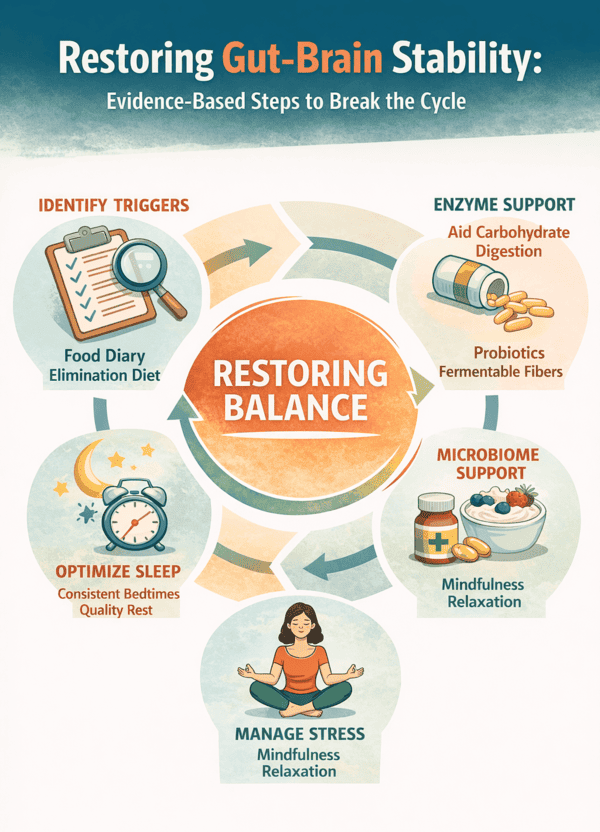
1. Identify Dietary Triggers Without Creating New Problems
For individuals with suspected FODMAP Sensitivity, clarity around dietary triggers is foundational. The low-FODMAP diet remains one of the most rigorously studied dietary interventions for IBS and related symptoms, with consistent evidence supporting symptom reduction when implemented correctly.
However, the emphasis must be on structure and reintroduction — not indefinite restriction. The elimination phase is temporary. The reintroduction phase identifies individual tolerance thresholds. When managed properly, this approach narrows rather than broadens restriction.
Working with a registered dietitian is strongly advisable. Unsupervised elimination diets can drift toward unnecessary food avoidance, increasing anxiety around eating and raising the risk of nutritional imbalance. The goal is strategic identification of triggers, not dietary hypervigilance.
2. Strategic Enzyme Support
For some individuals, strict avoidance proves socially and nutritionally burdensome. In these cases, targeted enzyme supplementation can serve as a pragmatic bridge.
Lactase supplementation is well established for lactose intolerance. Alpha-galactosidase can reduce fermentation from certain legumes and vegetables. Enzymes formulated to assist with fructan and GOS digestion may provide benefit for some people with FODMAP Sensitivity.
These interventions are not curative. They do not “heal” the underlying sensitivity. But they can reduce symptom severity, allowing greater dietary flexibility and lowering the stress associated with social eating — which itself influences gut reactivity.
3. Support the Microbiome With Precision
A diverse, resilient microbiome buffers the gut–brain axis. Bacterial species that produce short-chain fatty acids such as butyrate help maintain intestinal barrier integrity and regulate inflammatory signaling.
Dietary fiber remains central, though the type and amount tolerated varies widely among individuals with FODMAP Sensitivity. Gradual reintroduction of tolerated fermentable fibers can promote microbial diversity without provoking excessive symptoms.
Probiotic research is nuanced. Certain strains have demonstrated benefit for specific IBS subtypes, but “probiotic” is not a blanket solution. Strain specificity matters. Dose matters. Clinical context matters. Random supplementation often yields inconsistent results.
For individuals with persistent symptoms, consulting a clinician familiar with microbiome-targeted strategies can prevent unnecessary experimentation.
4. Regulate the Stress Side of the Axis
Because the gut–brain axis runs in both directions, psychological stress meaningfully alters digestive physiology. Cortisol influences gut permeability. Sympathetic activation alters motility. Heightened vigilance increases pain perception.
Mindfulness-based stress reduction (MBSR) has demonstrated measurable improvements in IBS symptom severity. Cognitive behavioral approaches can reduce symptom-related anxiety and catastrophic interpretation of bodily sensations. Regular physical activity improves vagal tone and supports more stable gut motility.
Importantly, this is not about dismissing digestive symptoms as “stress.” It is about recognizing that stress physiology amplifies existing gut sensitivity. Addressing both sides of the axis produces better outcomes than focusing on one alone.
5. Use Sleep as a Regulatory Lever
Sleep is often treated as an afterthought in digestive health. It should not be.
Circadian rhythms regulate gastrointestinal motility, microbial composition, and epithelial barrier function. Sleep deprivation increases gut permeability and inflammatory signaling. For individuals already managing FODMAP Sensitivity or related intolerances, chronic poor sleep compounds dysregulation.
Consistent sleep timing, light exposure management, and minimizing late-night eating can stabilize both neurological and digestive rhythms. It is unglamorous advice, but its physiological impact is substantial.
Final Thoughts
Digestive symptoms are often minimized — by patients, by clinicians, and by healthcare systems that separate gastroenterology from mental health. Bloating is tolerated. Cramping is normalized. Brain fog is attributed to stress. Anxiety is treated in isolation. Meanwhile, the gut continues signaling.
FODMAP Sensitivity, and related intolerances, do not exist in a vacuum. Repeated fermentation, inflammation, and visceral hypersensitivity create a physiological environment that can destabilize mood, sharpen stress responses, and erode cognitive clarity. When the gut is chronically dysregulated, the nervous system adapts — often toward vigilance.
This does not mean every emotional symptom originates in the digestive tract. It does mean that for a meaningful subset of individuals, especially those living with IBS or unexplained digestive volatility, the gut–brain axis deserves careful attention. Treating anxiety without evaluating dietary triggers may leave a perpetuating factor intact. Likewise, treating digestion without addressing stress physiology may limit progress.
The goal is not dietary rigidity or supplement dependence. It is precision. Identifying what the gut tolerates, stabilizing neural signaling, and restoring rhythm to digestion and sleep can meaningfully shift how the brain experiences the world.
Before assuming your mind is malfunctioning, it may be worth asking whether your gut has been trying to compensate for something it cannot process.
That question alone changes the frame.